Descripción
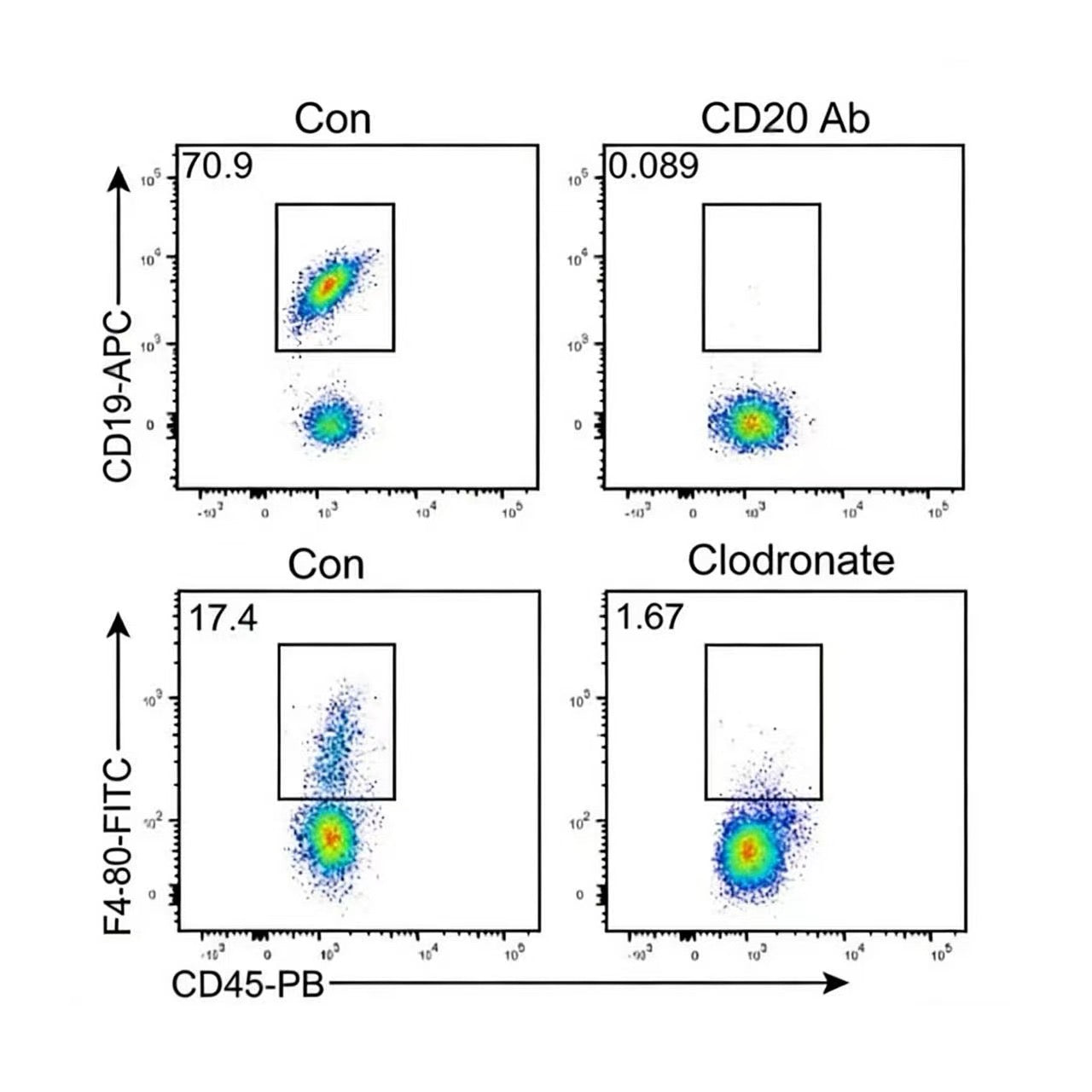
Clodronate Liposomes are currently the most mature, convenient, and cost-effective tool for macrophage depletion. They can effectively eliminate macrophages in various tissues and sites within animals, including the liver, spleen, lungs, and blood, and are currently the most widely used method for macrophage depletion.
After Clodronate Liposomes are injected into a living organism, they are phagocytosed by macrophages. Under the action of lysosomal phosphatases within the macrophages, the clodronate dissolved in the liposomes is gradually released and accumulates intracellularly. When a certain concentration is reached, the macrophages will suffer irreversible damage, inducing apoptosis.
This product is a combination pack, containing an In Vivo Macrophage Depletion Reagent (Cat#40337ES) and an Empty Liposome Control (Cat#40338ES).
Features
Efficient Macrophage Depletion: Efficient macrophage depletion through clodronate-encapsulated liposomes.
High specificity: Selectively targeting phagocytic macrophages and monocytes.
Validated in Multiple Models: Validated in multiple animal models for immunology, inflammation, and tumor microenvironment studies.
Ready-to-Use Format: Ready-to-use formulation for convenient and reproducible experiments.
Components
|
Components No. |
Name |
Cat.No. |
Storage |
||
|
40339ES05 |
40339ES08 |
40339ES10 |
|||
|
40337ES |
Clodronate Liposomes |
2 mL |
5 mL |
10 mL |
2~8℃ |
|
40338ES |
Control Liposomes(PBS) |
2 mL |
5 mL |
10 mL |
2~8℃ |
Storage
This product should be stored at 2~8°C. Valid for 6 months. Do not freeze!
Figure
1. Case Study: Efficient Depletion of Perivascular Macrophages in the Mouse Brain

Figure 1. Efficient depletion of perivascular macrophages in the brain following intracisternal administration of Yeasen Macrophage Depletion Reagent (Cat. No. 40337ES).
Mice received a single injection of 4 µL clodronate liposomes (CLO) into the cisterna magna; control animals received an equal volume of PBS. Brain tissues were collected 7 days post-injection and sectioned for immunofluorescence staining using anti-CD163 antibody (Cell Signaling Technology, #24595) to label perivascular macrophages. Quantification of macrophages per unit vessel length showed a significant reduction in the CLO group (33.10 ± X.XX) compared to PBS controls (96.59 ± X.XX), corresponding to a ~66% decrease (or approximately 34% of control levels). Note: If CD26 was intended instead of CD163, please confirm—CD163 is a canonical marker for perivascular macrophages in the CNS; CD26 is atypical for this population.
Documents:
Safety Data Sheet
Manuals
Related Blog
Clodronate Liposomes | Lesson 1: Injection Methods and Administration Strategies
Clodronate Liposomes | Lesson 2: Liver
Clodronate Liposomes | Lesson 3: Lung Macrophages
Clodronate Liposomes | Lesson 4: Intestine
Clodronate Liposomes | Lesson 5: Brain
Clodronate Liposomes | Lesson 6: Applications in C57BL/6 Mice
Clodronate Liposomes | Lesson 7: Peripheral Blood
Clodronate Liposomes | Lesson 8: Applications in Splenic Macrophage Depletion
Clodronate Liposomes | Lesson 9: Testicular Macrophage Depletion
Simplifying In Vivo Macrophage Depletion: A Practical Guide with Clodronate Liposomes
Macrophage Depletion Made Simple: A Comprehensive FAQ on Clodronate Liposomes
Macrophages and In Vitro/In Vivo Research Strategies
Macrophage Depletion Procedures and Literature Case Analysis
Try it for free!
Get a free sample of Clodronate Liposomes Kit and validate its performance in your own lab.
👉 [Request Sample Now]. Worldwide Shipping!

FAQ
Q1: For liver macrophage depletion, is perfusion required? Any recommendations for flow cytometry?
A1: Without perfusion, flow cytometry data is meaningless—macrophages won’t be properly isolated, making it impossible to assess differences. For flow cytometry, we recommend staining for F4/80 and CD11b. For immunohistochemistry, tail vein injection is recommended; harvest liver at 24 h post-injection. Note: Perfusion is not required for IHC.
Q2: Is the empty liposome control for the in vivo macrophage depletion reagent unilamellar or multilamellar?
A2: Multilamellar.
Q3: What is the concentration of the Macrophage Depletion Reagent?
A3: 5 mg/mL.
Q4: What is the particle size of Clodronate Liposomes (in vivo macrophage depletion reagent)?
A4: 150 nm – 3 μm.
Q5: How should Clodronate Liposomes be stored and handled after receipt?
A5: If not used immediately, store at 4°C. Do not freeze! Use as supplied—do not dilute. Liposomes may sediment over time; gently mix before use. Do not inject directly from 4°C—allow the suspension to equilibrate to room temperature before administration.
Q6: Can Clodronate Liposomes be used for in vitro macrophage depletion?
A6: Yes, they can be used in vitro, but this application is better suited for in vivo experiments. In vitro, clodronate released from dead cells or leaked from liposomes accumulates in the culture medium. Although free clodronate cannot cross intact cell membranes, prolonged exposure in culture may eventually lead to cellular uptake and non-specific toxicity. In contrast, in vivo, free clodronate is rapidly cleared by the kidneys and has a short half-life.
Q7: Animals died shortly after intravenous injection of Clodronate Liposomes—why?
A7: Two likely causes:
Injection of a non-homogeneous suspension—liposomes sediment over time. Always gently mix before and during use, especially when injecting multiple animals sequentially.
Injection of cold liposomes directly from 4°C—always warm to room temperature before administration.
Q8: For depleting macrophages in different organs/tissues, what injection route, dose, timing, and frequency should be used?
A8: This depends on the specific experimental design. We recommend that users develop their own protocol based on published literature relevant to their research goals. We can provide reference papers upon request.
Q9: Animals died several days after Clodronate Liposome injection—why?
A9: This may be due to opportunistic infections. Macrophage depletion compromises innate immunity, increasing susceptibility to bacteria, viruses, or fungi—especially if aseptic techniques were not strictly followed.
Q10: After IV injection of Clodronate Liposomes, ED1+ cells in rat spleen/liver were not fully depleted—why?
A10: In rats, mature macrophages are ED1+/ED2+, while some immature or less phagocytic precursors are ED1+/ED2–. Clodronate liposomes efficiently deplete ED2+ (mature, phagocytic) macrophages, but ED1+ precursors lacking phagocytic activity are not cleared. Thus, residual ED1+ cells reflect this precursor population.
Q11: Clodronate Liposomes did not achieve expected depletion efficiency—why?
A11: All batches are rigorously tested for clodronate concentration and contaminants before release. However, liposomes are sensitive to temperature extremes. Store and transport at 4–8°C. Do not freeze or expose to >30°C. Use within 3 months of receipt to ensure optimal performance.
Q12: Can the injection volume be increased during IV administration?
A12: For IV injection, volume should not exceed 0.1 mL per 10 g body weight. Larger volumes may be used for intraperitoneal (IP) injection. For subcutaneous (SC) injection, volume depends on the capacity of the injection site.
Q13: The Clodronate Liposomes froze during shipping—can they still be used?
A13: No. Do not use if frozen.
Q14: Are the Clodronate Liposomes anionic?
A14: Yes, they are anionic.
Q15: On the product datasheet, does “5 mg/mL” refer to the drug concentration?
A15: Yes, it refers to the clodronate concentration.
Citations & References:
- Xiong X, Chen S, Shen J, et al. Cannabis suppresses antitumor immunity by inhibiting JAK/STAT signaling in T cells through CNR2. Signal Transduct Target Ther. 2022;7(1):99. Published 2022 Apr 6. doi:10.1038/s41392-022-00918-y(IF:52.7)
- Jiang Z, Wang Y, Gong J, et al. An Aeromonas variant that produces aerolysin promotes susceptibility to ulcerative colitis. Science. 2025;390(6775):eadz4712. doi:10.1126/science.adz4712(IF:45.8)
- Wang Y, Chai Y, Liu Y, et al. Inhibition of tumor cell macropinocytosis driver DHODH reverses immunosuppression and overcomes anti-PD1 resistance. Immunity. 2025;58(10):2456-2471.e6. doi:10.1016/j.immuni.2025.07.013(IF:26.3)
- Chen S, Zhang P, Zhu G, et al. Targeting GSDME-mediated macrophage polarization for enhanced antitumor immunity in hepatocellular carcinoma. Cell Mol Immunol. 2024;21(12):1505-1521. doi:10.1038/s41423-024-01231-0(IF:19.8)
- Zhang Z, Chen C, Yang F, et al. Itaconate is a lysosomal inducer that promotes antibacterial innate immunity [published online ahead of print, 2022 May 25]. Mol Cell. 2022;S1097-2765(22)00443-9. doi:10.1016/j.molcel.2022.05.009(IF:17.970)
- Cai J, Peng J, Zang X, et al. Mammary Leukocyte-Assisted Nanoparticle Transport Enhances Targeted Milk Trace Mineral Delivery [published online ahead of print, 2022 Jun 30]. Adv Sci (Weinh). 2022;e2200841. doi:10.1002/advs.202200841(IF:17.521)
- Liu S, Zhang Z, Wang Z, et al. SPP1 Drives Colorectal Cancer Liver Metastasis and Immunotherapy Resistance by Stimulating CXCL12 Production in Cancer-Associated Fibroblasts. Cancer Res. 2026;86(1):58-79. doi:10.1158/0008-5472.CAN-24-4916(IF:16.6)
- Dai P, Sun Y, Huang Z, et al. USP2 inhibition unleashes CD47-restrained phagocytosis and enhances anti-tumor immunity. Nat Commun. 2025;16(1):4564. Published 2025 May 16. doi:10.1038/s41467-025-59621-5(IF:15.7)
- Jin H, Liu K, Tang J, et al. Genetic fate-mapping reveals surface accumulation but not deep organ invasion of pleural and peritoneal cavity macrophages following injury. Nat Commun. 2021;12(1):2863. Published 2021 May 17. doi:10.1038/s41467-021-23197-7(IF:14.919)
- Sheng D, Ma W, Zhang R, et al. Ccl3 enhances docetaxel chemosensitivity in breast cancer by triggering proinflammatory macrophage polarization [published correction appears in J Immunother Cancer. 2022 Jun;10(6):]. J Immunother Cancer. 2022;10(5):e003793. doi:10.1136/jitc-2021-003793(IF:13.751)
- Pan J, Zhang M, Rao D, et al. CAD manipulates tumor intrinsic DHO/UBE4B/NF-κB pathway and fuels macrophage cross-talk, promoting HCC metastasis. Hepatology. Published online March 12, 2025. doi:10.1097/HEP.0000000000001304(IF:13)
- Jia J, Ji W, Xiong N, Lin J, Yang Q. Trained immunity using probiotics and inactivated pathogens enhances resistance to Salmonella enterica serovar Typhimurium infection by activating the cGAS-STING signal pathway in mice and chickens. J Adv Res. 2026;79:491-504. doi:10.1016/j.jare.2025.03.011(IF:13)
- Lv Q, Yang H, Xie Y, et al. Prunus mume derived extracellular vesicle-like particles alleviate experimental colitis via disrupting NEK7-NLRP3 interaction and inflammasome activation. J Nanobiotechnology. 2025;23(1):532. Published 2025 Jul 21. doi:10.1186/s12951-025-03567-9(IF:12.6)
- Zhao L, Zhang H, Liu X, et al. TGR5 deficiency activates antitumor immunity in non-small cell lung cancer via restraining M2 macrophage polarization. Acta Pharm Sin B. 2022;12(2):787-800. doi:10.1016/j.apsb.2021.07.011(IF:11.614)
- Xia L, Zhang C, Lv N, et al. AdMSC-derived exosomes alleviate acute lung injury via transferring mitochondrial component to improve homeostasis of alveolar macrophages. Theranostics. 2022;12(6):2928-2947. Published 2022 Mar 21. doi:10.7150/thno.69533(IF:11.556)
- Zhang X, Hou L, Li F, et al. Piezo1-mediated mechanosensation in bone marrow macrophages promotes vascular niche regeneration after irradiation injury. Theranostics. 2022;12(4):1621-1638. Published 2022 Jan 16. doi:10.7150/thno.64963(IF:11.556)
- Wang H, Li L, Li Y, et al. Intravital imaging of interactions between iNKT and kupffer cells to clear free lipids during steatohepatitis. Theranostics. 2021;11(5):2149-2169. Published 2021 Jan 1. doi:10.7150/thno.51369(IF:11.556)
- Xun J, Du L, Gao R, et al. Cancer-derived exosomal miR-138-5p modulates polarization of tumor-associated macrophages through inhibition of KDM6B. Theranostics. 2021;11(14):6847-6859. Published 2021 May 3. doi:10.7150/thno.51864(IF:11.556)
- Zuo L, Li J, Zhang X, et al. Aberrant mesenteric adipose extracellular matrix remodeling is involved in adipocyte dysfunction in Crohn's disease: The role of TLR-4-mediated macrophages [published online ahead of print, 2022 Jun 16]. J Crohns Colitis. 2022;jjac087. doi:10.1093/ecco-jcc/jjac087(IF:9.071)
- Huang C, Wang J, Liu H, et al. Ketone body β-hydroxybutyrate ameliorates colitis by promoting M2 macrophage polarization through the STAT6-dependent signaling pathway. BMC Med. 2022;20(1):148. Published 2022 Apr 15. doi:10.1186/s12916-022-02352-x(IF:8.775)
- Jiang P, Gao W, Ma T, et al. CD137 promotes bone metastasis of breast cancer by enhancing the migration and osteoclast differentiation of monocytes/macrophages. Theranostics. 2019;9(10):2950-2966. Published 2019 May 9. doi:10.7150/thno.29617(IF:8.063)
- Yang XL, Wang G, Xie JY, et al. The Intestinal Microbiome Primes Host Innate Immunity against Enteric Virus Systemic Infection through Type I Interferon. mBio. 2021;12(3):e00366-21. Published 2021 May 11. doi:10.1128/mBio.00366-21(IF:7.867)
- Sun Z, Huang W, Zheng Y, et al. Fpr2/CXCL1/2 Controls Rapid Neutrophil Infiltration to Inhibit Streptococcus agalactiae Infection. Front Immunol. 2021;12:786602. Published 2021 Nov 24. doi:10.3389/fimmu.2021.786602(IF:7.561)
- Cai J, Cui X, Wang X, You L, Ji C, Cao Y. A Novel Anti-Infective Peptide BCCY-1 With Immunomodulatory Activities. Front Immunol. 2021;12:713960. Published 2021 Jul 22. doi:10.3389/fimmu.2021.713960(IF:7.561)
- Li C, Song J, Guo Z, et al. EZH2 Inhibitors Suppress Colorectal Cancer by Regulating Macrophage Polarization in the Tumor Microenvironment. Front Immunol. 2022;13:857808. Published 2022 Apr 1. doi:10.3389/fimmu.2022.857808(IF:7.561)
- Tian L, Li W, Yang L, et al. Cannabinoid Receptor 1 Participates in Liver Inflammation by Promoting M1 Macrophage Polarization via RhoA/NF-κB p65 and ERK1/2 Pathways, Respectively, in Mouse Liver Fibrogenesis. Front Immunol. 2017;8:1214. Published 2017 Sep 28. doi:10.3389/fimmu.2017.01214(IF:6.429)
- Wang X, Li W, Jiang H, et al. Zebrafish Xenograft Model for Studying Pancreatic Cancer-Instructed Innate Immune Microenvironment. Int J Mol Sci. 2022;23(12):6442. Published 2022 Jun 9. doi:10.3390/ijms23126442(IF:5.924)
- Xiong C, Zhu Y, Xue M, et al. Tumor-associated macrophages promote pancreatic ductal adenocarcinoma progression by inducing epithelial-to-mesenchymal transition. Aging (Albany NY). 2021;13(3):3386-3404. doi:10.18632/aging.202264(IF:5.682)
- Wang J, Li X, Wang Y, Li Y, Shi F, Diao H. Osteopontin aggravates acute lung injury in influenza virus infection by promoting macrophages necroptosis. Cell Death Discov. 2022;8(1):97. Published 2022 Mar 4. doi:10.1038/s41420-022-00904-x(IF:5.241)
- Ma Y, Liang Y, Wang N, et al. Avian Flavivirus Infection of Monocytes/Macrophages by Extensive Subversion of Host Antiviral Innate Immune Responses. J Virol. 2019;93(22):e00978-19. Published 2019 Oct 29. doi:10.1128/JVI.00978-19(IF:5.103)
-
Jiang Q, Li W, Zhu X, et al. Estrogen receptor β alleviates inflammatory lesions in a rat model of inflammatory bowel disease via down-regulating P2X7R expression in macrophages. Int J Biochem Cell Biol. 2021;139:106068. doi:10.1016/j.biocel.2021.106068(IF:5.085)
Pago y seguridad
Su información de pago se procesa de forma segura. No almacenamos detalles de la tarjeta de crédito ni tenemos acceso a la información de su tarjeta de crédito.
También te puede gustar
Consulta
Preguntas frecuentes
El producto es solo para fines de investigación y no está destinado a uso terapéutico o diagnóstico en humanos o animales. Los productos y el contenido están protegidos por patentes, marcas comerciales y derechos de autor propiedad de Yeasen Biotechnology. Los símbolos de marca comercial indican el país de origen, no necesariamente el registro en todas las regiones.
Algunas aplicaciones pueden requerir derechos de propiedad intelectual adicionales de terceros.
Yeasen se dedica a la ciencia ética y cree que nuestra investigación debe abordar cuestiones críticas al tiempo que garantiza la seguridad y los estándares éticos.