Description
Eukaryotes mRNA forms a special structure at the 5'end after transcription, which is the cap structure. The cap structure plays an important role in the stability, transportation, and translation of mRNA. The vaccinia virus capping enzyme is an effective enzyme that can catalyze the formation of the cap structure. It"s composed of two subunits D1 and D12. It also has RNA triphosphatase activity, guanylate acyltransferase activity, and guanine methyltransferase activity that could connect the 7-methylguanine cap structure (m7Gppp) to the 5'end of the RNA (m7Gppp5'N). Vaccinia virus capping enzyme can cap the RNA in correct direction within one hour when present at a suitable concentration of capping buffer, guanosine triphosphate (GTP), S-adenosylmethionine (SAM), etc.
Feature
- Isolated from a recombinant source
- Tested for the absence of endonucleases, exonucleases, RNases
Application
- Capping mRNA before translation assays/in vitro translation
- Labeling 5´ end of mRNA
Specification
| Source | Recombinant E. coli with vaccinia virus capping enzyme gene |
| Optimum Temperature | 37℃ |
| Storage Buffer | 20 mM Tris-HCl pH 8.0, 100 mM NaCl, 1 mM DTT, 0.1 mM EDTA, 0.1% Triton X-100, 50% glycerin |
| Unit Definition | 1 unit: The amount of enzyme required to incorporate 10 pmol GTP (α- 32P) into a transcript with 80 nucleotides (80 nt) at 37℃ within 1 hour. |
Components
| Components No. | Name | 10615ES84 (2,000 U) | 10615ES92 (10,000 U) | 10615ES96 (100,000 U) | 10615ES99 (5 MU) |
| 10615 | mRNA Vaccinia Capping Enzyme (10 U/μL) | 200 μL | 1 mL | 10 mL | 500 mL |
Shipping and Storage
mRNA Vaccinia Capping Enzyme GMP-grade products are shipped with dry ice and can be stored at -15℃ ~ -25℃ for one year.
Figures
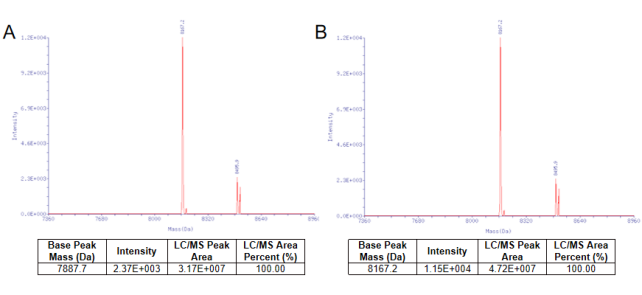
Figure 1. The capping efficiency almost reached 100% by the vaccinia capping system from YEASEN. Nucleic acid mass spectrometry before the cap reaction was shown in Figure 1A. Nucleic acid mass spectrometry after the cap reaction was shown in Figure 1B.
FAQ
Q: What is the purpose of heating first and then cooling down in the enzymatic cap addition process? It seems quite difficult to achieve heating followed by cooling during large-scale production.
A: The purpose of preheating is to unfold the secondary structure of RNA and bring it to a single-stranded state. If this step is not carried out, it will affect the efficiency of capping. This part is indeed a current technical challenge in production. Some companies now directly predict the secondary structure of RNA when determining the template sequence. The capping enzyme only recognizes the first few nucleotides of mRNA. If it is determined that this structure will not result in the secondary structure of RNA, then heating can be omitted.
Q: How to choose between cap-like analogues and enzymatic cap addition?
A: In industry, the most commonly used method for enzymatic cap addition is to treat IVT products with vaccinia virus cap-adding enzyme, which can modify them into Cap 0 mRNA. The Cap 0 structure can be further modified into Cap 1 (m7GpppmN) under the action of 2'O-methyltransferase. With enzymatic cap addition, the cap addition efficiency can reach over 95%. The co-transcription cap addition method is simple to operate, but because GTP competes with the cap dimer, the cap addition rate of this method is a little lower; both methods have their own advantages and disadvantages.
Q: What are the methods for optimizing the cap?
A: By increasing the amount of VCE, for example, adding 10 ul of 100 U of enzyme activity to a 100 ul system, the main purpose is to enhance the methyltransferase activity of VCE. It can also be achieved by increasing SAM (0.5 mmol) to enhance the guanine transfer activity and increase the cap formation rate. For instance, our new single-strand cap enzyme has a significantly improved cap formation rate. This is because its guanine transfer activity is better than that of traditional VCE. For traditional VCE, the small subunit D12 is prone to precipitation and polymer formation, which makes purification difficult and leads to loss, thus resulting in higher costs and inability to control batches.
Q: For some mRNAs that do not have caps added, what impact will this have on subsequent experiments, such as those involving the production of biological agents?
A: Currently, the industry has not specifically targeted this aspect. In the industry, the main focus is on the efficiency of the cap application. The current best cap application rate is above 80%. Of course, the higher the better. There is still no absolute standard for this.
For the synthesized mRNA, purification methods can be employed, such as the one-step membrane encapsulation method by Kang*. This significantly reduces the purification process. The traditional method uses Oligo d(T) columns for purification, with good results achieving a recovery rate of 70-80%; poor results have a recovery rate of 40-50%. An additional step of a hydrophobic column can also be added after the Oligo d(T) column.
Q: What are the requirements for the starting position of the template for co-transcriptional cap addition and two-step cap addition?
A: The co-transcription requirement stipulates that the starting position of the template must begin with AGG*.
The two-step method does not have any specific requirements for the position. However, our current products have the highest yield for the AGGG starting position, mainly due to the preference of the T7 enzyme.
Documents
Payment & Security
Your payment information is processed securely. We do not store credit card details nor have access to your credit card information.
You may also like
Inquiry
FAQ
The product is for research purposes only and is not intended for therapeutic or diagnostic use in humans or animals. Products and content are protected by patents, trademarks, and copyrights owned by Yeasen Biotechnology. Trademark symbols indicate the country of origin, not necessarily registration in all regions.
Certain applications may require additional third-party intellectual property rights.
Yeasen is dedicated to ethical science, believing our research should address critical questions while ensuring safety and ethical standards.