Description
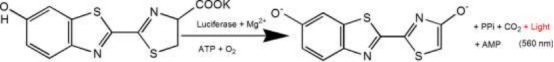
D-luciferin is a common substrate for Luciferase and is widely used throughout biotechnology, especially in vivo imaging Technology. The mechanism of action is that luciferin (substrate) can be oxidized to emit light in the presence of ATP and luciferase. When fluorescein is in excess, the quantum number of light produced is equal to Luciferase concentrations were positively correlated (see figure below). Plasmids carrying luciferase encoding gene (Luc) were transfected into cells and introduced into study animals such as rats and mice.
D-luciferin is also commonly used in in vitro studies, including luciferase and ATP levels analysis; Reporter gene analysis; High-throughput sequencing and various contamination tests. There are three Product form: D-luciferin (free acid), D-luciferin salt (sodium salt and potassium salt). The main difference lies in solubility: water solubility of the former and solubility of the buffer system. They are weakly soluble except in weak bases such as low concentrations of NaOH and KOH solutions. Soluble in methanol and DMSO; The latter can be easily dissolved in water or buffer, easy to use.
Features
- No radiation, almost harmless to living organisms.
- Bioluminescence, no excitation light source.
- So sensitive, you can detect it in a few hundred cells.
- Good penetration, 3-4cm tissue depth can still be detected.
- High signal-to-noise ratio, strong fluorescence signal and good anti-interference.
Applications
- In the tumorigenesis experiment in nude mice, the tumor growth was observed without invasion in real time, without tumor stripping measurement.
- To test the effect of the administration on tumor growth or metastasis, the fluorescein substrate can be completely eliminated within 3 hours, without interference to the drug experiment.
- The localization and distribution of foreign cells in animals were detected.
- The target gene or promoter of the target gene is fused to the luciferase gene to detect changes in gene expression during drug treatment or disease progression.
- Monitoring stem cell transplantation, survival and proliferation; Trace the distribution and migration of stem cells in vivo.
Specifications
| English synonym | (S)-4,5-Dihydro-2-(6-hydroxy-2-benzothiazolyl)-4-thiazolecarboxylic acid potassium salt; D-Luciferin firefly, potassium salt |
| CAS NO. | 115144-35-9 |
| Formula | C11H7N2O3S2K |
| Molecular weight | 318.42 g/mol |
| Appearance | Light yellow powder |
| Solubility | Soluble in water(60 mg/mL) |
Components
| Components No. | Name | 40902ES01 | 40902ES02 | 40902ES03 | 40902ES08 |
| 40902 | D-Luciferin, Potassium Salt | 100mg | 500 mg | 1g | 5g |
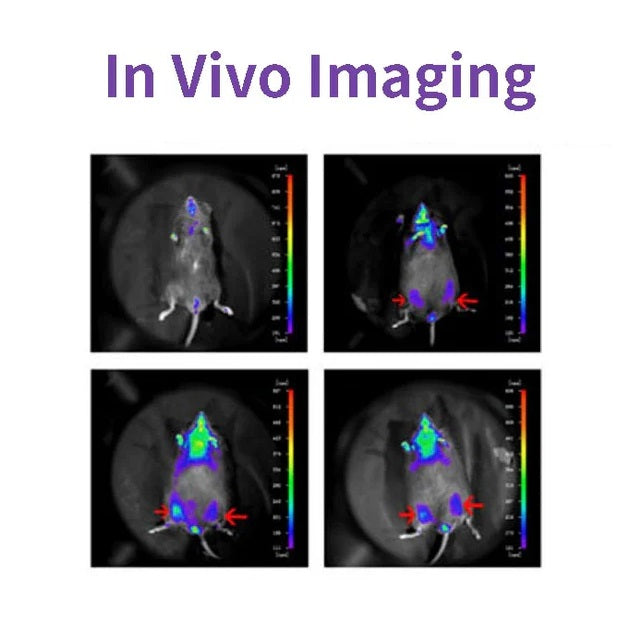
Figures

Cited from "A Novel Allosteric Inhibitor of Phosphoglycerate Mutase 1 Suppresses Growth and Metastasis of Non-Small-Cell Lung Cancer, Cell Metab. 2019;30(6):1107-1119
Shipping and Storage
The product is shipped with dry ice and can be stored at -15℃ ~ -25℃ for 1 year.
FAQ
Q: Are there any differences between Firefly Luciferin, Beetle Luciferin and D-Luciferin?
A: There is no difference. The three are merely variations in naming by different companies, and they are all compounds (S)-2-(6-Hydroxy-2-benzothiazolyl)-2 -thiazoline-4-carboxylic acid
Q: What applications do this series of products mainly serve?
A: Besides being used for in vivo imaging, fluorescent dye products are also employed in other applications involving luciferase, such as in vitro reporter gene assays, microbial/viral monitoring, pyrosequencing, etc.
Q: What is the difference between sodium fluorescein and D-fluorescein sodium?
A: Sodium fluorescein: It can be used for cell permeability testing. The permeability of the pigment can be determined by measuring the absorbance value at OD490. D-fluorescein sodium salt: Used for in vivo imaging and reporter gene systems. It is detected through a cold luminescence module and does not require excitation light stimulation.
Q: What are the differences between potassium salt, sodium salt and free acid of fluorescein?
The main differences among the three are as follows: 1) Solubility: The salt form is easily soluble in water. The solubility of potassium salt is 60mg/ml, and that of sodium salt is 100mg/ml. Free acid is not easily soluble in water. It can be dissolved by a weak alkaline solution of sodium bicarbonate, and its solubility in methanol is 10mg/ml, and in DMSO is 50mg/ml. 2) Toxicity: The salt form is more convenient to use during the process, especially in in vivo imaging experiments, as it can dissolve in water and the reaction toxicity will be smaller (Fluorescein is a low-molecular-weight organic compound composed of phenylthiazole and thiazole carboxyl group, with low toxicity). 3) Usage effect: There is no significant difference. In in vivo experimental studies, the usage rate of potassium salt is higher.
Q: Does the purity of the fluorescent dye have any impact on the experiment?
A: It has an impact. The purity is above 99%, with a good level of 99%. For 99% pure fluorescein, there is 1% solid impurity. If this 1 gram of impurity is dissolved in 25 ml of buffer solution (this dilution ratio is the standard dilution method for in vivo imaging experiments), the concentration of the impurity at this time is 0.4 g/L. Assuming the molecular weight of the impurity is 1000 g/mol, then the molar concentration of the impurity is 400 μM. This concentration may inhibit the action of certain enzymes in the cells and may reduce the experimental effect or cause harm to the animals.
Q: How stable is the product?
A: The powder should be stored in a dark place at -20 or -70 degrees Celsius. Its validity period is at least one year.
Q: How does the substrate for in vivo imaging work?
A: D-fluorescein (D-Luciferin) is a commonly used substrate for luciferase. Its mechanism of action is that under the action of ATP and luciferase, the fluorescent substance (the substrate) can be oxidized and emit light. When there is an excess of fluorescent substance, the number of photons produced is positively correlated with the concentration of luciferase. 2. Native coelenterazine is an action substrate for various luciferases such as sea kidney luciferase (Rluc) and Gaussia luciferase (Gluc).
Q: Are there any recommended instruments? Can the multi-functional microplate reader be used?
A: Recommended instruments: 1. Instruments with a biochemiluminescence detection module. The light produced by luciferin can be detected by a photometer or a scintillation counter. Common in vivo imaging instruments: such as the IVIS® Lumina small animal in vivo imaging system, the In-Vivo Xtreme multi-mode small animal in vivo imaging instrument from Bruker, Germany. 2. Multi-functional microplate readers: It is necessary to confirm with the instrument manufacturer whether it is a specific biochemiluminescence detection module (note: cannot use a fluorescence microscope).
Q: Can fluorescein penetrate the cell membrane?
A: Fluorescein is a small molecule with excellent water solubility and lipid solubility. It can easily penetrate the cell membrane and the blood-brain barrier.
Q: Which is better, tail vein injection or intraperitoneal injection?
A: Either one is fine. Intraperitoneal injection has a slower diffusion rate and the light emission lasts longer; intravenous injection has a faster diffusion rate, but the light emission lasts for a very short period of time.
Q: How many mice can 100mg be injected into?
A: Intraperitoneal injection (i.p.), with a fluorescein concentration of 150 mg/kg per kg of body weight. Taking a 20g C57 mouse as an example, the dosage for each mouse for one test: 150 mg/kg × 20g = 3mg, 100mg ÷ 3mg = 33 mice. This is for reference only. The specific dosage should be based on the reagent and without any loss.
Q: Is potassium d-fluorescein toxic to animals?
A: Generally speaking, potassium d-fluorescein does not cause any toxic effects on animals.
Q: What is the decomposition rate of potassium salt of fluorescein?
A: There is no such information.
Q: The process of fluorescent substance oxidation and luminescence requires magnesium ions and ATP. If D-fluorescein potassium salt is used for in vitro imaging, how can ATP and magnesium ions be provided? It is recommended to dissolve the potassium salt in distilled water for in vitro use.
A: This product undergoes in vitro imaging. The ATP and magnesium ions required for this process need to be achieved through the cell culture medium. Therefore, a cell culture medium containing ATP and magnesium ions needs to be added to dilute the probe; 2. The cells themselves also contain a certain amount of ATP and magnesium, which can promote the reaction. In most cases, the ATP and magnesium contained in the cells are sufficient for the reaction of D-fluorescein salt, but the content varies depending on the type of cells. You can first try using a culture medium without additional ATP and magnesium, and conduct the experiment normally to see the results.
Q: What are the excitation wavelength and emission wavelength of the substrate?
A: Chemiluminescence, no excitation wavelength required. The detection wavelength for firefly luciferin is 560 nm. The detection wavelength for sea kidney luciferin is 465 nm.
Q: Can the substrate enter living cells?
A: It can pass through the blood-brain barrier, the placental barrier and the blood test barrier, and also enter living cells.
Q: Regarding the injection method and dosage of fluorescent substances?
A: The recommended concentrations are derived from the literature: 1) Injection method: It can be done through intraperitoneal injection or tail vein injection. 2) Injection volume: The scientific approach is to evaluate the injection dose based on the kinetic curve. The initial attempted injection dose is: 150mg substrate/kg of mouse body weight. Therefore, the purchase quantity can be calculated according to the above method: If there are 10 mice, 22 - 25g, then 33 - 37.5mg of substrate is required.
Q: How does the luminescence property of fluorescein work?
A: Approximately 3 minutes after intraperitoneal injection of fluorescein into mice, cells expressing luciferase began to emit light. The intensity reached a stable peak after 10 minutes, and this peak lasted for about 10 - 15 minutes before gradually diminishing. Detection can be conducted within 10 - 15 minutes after the injection. This is for reference only. It is recommended to conduct a preliminary experiment to establish the kinetics curve of luciferase, thereby determining the optimal time for signal detection and the signal plateau period.
Q: What are the reasons for the lack of effect in the live imaging experiment?
A: To successfully conduct the in vivo imaging experiment, the following conditions are required: The target tissue or cells must express the luciferase gene; the injection of the luciferin substrate must be successful; and the thickness of the tissue at the luminescent site is also a factor. If the experiment fails, the reasons can be investigated from these aspects: whether the luciferase gene is expressed, whether the luciferin substrate was injected correctly, and whether the luminescent site is deep.
Documents:
Citations & References:
[1] Gu Y, Wang Y, He L, et al. Circular RNA circIPO11 drives self-renewal of liver cancer initiating cells via Hedgehog signaling. Mol Cancer. 2021;20(1):132. Published 2021 Oct 14. doi:10.1186/s12943-021-01435-2(IF:27.401)
[2] Huang K, Liang Q, Zhou Y, et al. A Novel Allosteric Inhibitor of Phosphoglycerate Mutase 1 Suppresses Growth and Metastasis of Non-Small-Cell Lung Cancer [published correction appears in Cell Metab. 2021 Jan 5;33(1):223]. Cell Metab. 2019;30(6):1107-1119.e8. doi:10.1016/j.cmet.2019.09.014(IF:22.415)
[3] Dong X, Cheng R, Zhu S, et al. A Heterojunction Structured WO<sub>2.9</sub>-WSe<sub>2</sub> Nanoradiosensitizer Increases Local Tumor Ablation and Checkpoint Blockade Immunotherapy upon Low Radiation Dose. ACS Nano. 2020;14(5):5400-5416. doi:10.1021/acsnano.9b08962(IF:14.588)
[4] Zheng DW, Gao F, Cheng Q, et al. A vaccine-based nanosystem for initiating innate immunity and improving tumor immunotherapy. Nat Commun. 2020;11(1):1985. Published 2020 Apr 24. doi:10.1038/s41467-020-15927-0(IF:12.121)
[5] Hu J, Su H, Cao H, et al. AUXIN RESPONSE FACTOR7 integrates gibberellin and auxin signaling via interactions between DELLA and AUX/IAA proteins to regulate cambial activity in poplar. Plant Cell. 2022;34(7):2688-2707. doi:10.1093/plcell/koac107(IF:11.277)
[6] Qi P, Huang M, Hu X, et al. A Ralstonia solanacearum effector targets TGA transcription factors to subvert salicylic acid signaling. Plant Cell. 2022;34(5):1666-1683. doi:10.1093/plcell/koac015(IF:11.277)
[7] Huang X, Qiu M, Wang T, et al. Carrier-free multifunctional nanomedicine for intraperitoneal disseminated ovarian cancer therapy. J Nanobiotechnology. 2022;20(1):93. Published 2022 Feb 22. doi:10.1186/s12951-022-01300-4(IF:10.435)
[8] Tang Y, Lin S, Yin S, et al. In situ gas foaming based on magnesium particle degradation: A novel approach to fabricate injectable macroporous hydrogels. Biomaterials. 2020;232:119727. doi:10.1016/j.biomaterials.2019.119727(IF:10.273)
[9] Zhang X, Zheng S, Hu C, et al. Cancer-associated fibroblast-induced lncRNA UPK1A-AS1 confers platinum resistance in pancreatic cancer via efficient double-strand break repair. Oncogene. 2022;41(16):2372-2389. doi:10.1038/s41388-022-02253-6(IF:9.867)
[10] Qiao K, Liu Y, Xu Z, et al. RNA m6A methylation promotes the formation of vasculogenic mimicry in hepatocellular carcinoma via Hippo pathway. Angiogenesis. 2021;24(1):83-96. doi:10.1007/s10456-020-09744-8(IF:9.780)
[11] Wu Q, Kuang K, Lyu M, et al. Allosteric deactivation of PIFs and EIN3 by microproteins in light control of plant development. Proc Natl Acad Sci U S A. 2020;117(31):18858-18868. doi:10.1073/pnas.2002313117(IF:9.412)
[12] Meng DF, Sun R, Liu GY, et al. S100A14 suppresses metastasis of nasopharyngeal carcinoma by inhibition of NF-kB signaling through degradation of IRAK1. Oncogene. 2020;39(30):5307-5322. doi:10.1038/s41388-020-1363-8(IF:7.971)
[13] Gao Y, Wang J, Han H, et al. A nanoparticle-containing polycaprolactone implant for combating post-resection breast cancer recurrence. Nanoscale. 2021;13(34):14417-14425. Published 2021 Sep 2. doi:10.1039/d1nr04125h(IF:7.790)
[14] Jiang W, Li T, Guo J, et al. Bispecific c-Met/PD-L1 CAR-T Cells Have Enhanced Therapeutic Effects on Hepatocellular Carcinoma. Front Oncol. 2021;11:546586. Published 2021 Mar 10. doi:10.3389/fonc.2021.546586(IF:6.244)
[15] Zhou Y , Zhou C , Zou Y , et al. Multi pH-sensitive polymer-drug conjugate mixed micelles for efficient co-delivery of doxorubicin and curcumin to synergistically suppress tumor metastasis. Biomater Sci. 2020;8(18):5029-5046. doi:10.1039/d0bm00840k(IF:6.183)
[16] Zhao X, Liu X, Zhang P, et al. Injectable peptide hydrogel as intraperitoneal triptolide depot for the treatment of orthotopic hepatocellular carcinoma. Acta Pharm Sin B. 2019;9(5):1050-1060. doi:10.1016/j.apsb.2019.06.001(IF:5.808)
[17] Li M, Wang J, Yu Y, et al. Characterization of Mesenchymal Stem Cells Derived from Bisphosphonate-Related Osteonecrosis of the Jaw Patients' Gingiva. Stem Cell Rev Rep. 2022;18(1):378-394. doi:10.1007/s12015-021-10241-8(IF:5.739)
[18] Zhang X, Li Y, Ji J, et al. Gadd45g initiates embryonic stem cell differentiation and inhibits breast cell carcinogenesis. Cell Death Discov. 2021;7(1):271. Published 2021 Oct 2. doi:10.1038/s41420-021-00667-x(IF:5.241)
[19] Wu D, Lv J, Zhao R, et al. PSCA is a target of chimeric antigen receptor T cells in gastric cancer. Biomark Res. 2020;8:3. Published 2020 Jan 28. doi:10.1186/s40364-020-0183-x(IF:4.866)
[20] Qiao K, Chen C, Liu H, Qin Y, Liu H. Pinin Induces Epithelial-to-Mesenchymal Transition in Hepatocellular Carcinoma by Regulating m6A Modification. J Oncol. 2021;2021:7529164. Published 2021 Dec 7. doi:10.1155/2021/7529164(IF:4.375)
[21] Li K, Liu T, Chen J, Ni H, Li W. Survivin in breast cancer-derived exosomes activates fibroblasts by up-regulating SOD1, whose feedback promotes cancer proliferation and metastasis. J Biol Chem. 2020;295(40):13737-13752. doi:10.1074/jbc.RA120.013805(IF:4.238)
[22] Kong Y, Feng Z, Chen A, et al. The Natural Flavonoid Galangin Elicits Apoptosis, Pyroptosis, and Autophagy in Glioblastoma. Front Oncol. 2019;9:942. Published 2019 Sep 27. doi:10.3389/fonc.2019.00942(IF:4.137)
[23] Kong Y, Feng Z, Chen A, et al. The Natural Flavonoid Galangin Elicits Apoptosis, Pyroptosis, and Autophagy in Glioblastoma. Front Oncol. 2019;9:942. Published 2019 Sep 27. doi:10.3389/fonc.2019.00942(IF:4.137)
[24] Hu J, Wang Y, Yuan Y. Inhibitors of APE1 redox function effectively inhibit γ-herpesvirus replication in vitro and in vivo. Antiviral Res. 2021;185:104985. doi:10.1016/j.antiviral.2020.104985(IF:4.101)
[25] Li Q, Wei D, Feng F, et al. α2,6-linked sialic acid serves as a high-affinity receptor for cancer oncolytic virotherapy with Newcastle disease virus. J Cancer Res Clin Oncol. 2017;143(11):2171-2181. doi:10.1007/s00432-017-2470-y(IF:3.503)
[26] Han B, Jiang P, Liu W, et al. Role of Daucosterol Linoleate on Breast Cancer: Studies on Apoptosis and Metastasis. J Agric Food Chem. 2018;66(24):6031-6041. doi:10.1021/acs.jafc.8b01387(IF:3.412)
[27] Hu H, Zhang Z, Wang R, et al. BGC823 Cell Line with the Stable Expression of iRFP720 Retains Its Primary Properties with Promising Fluorescence Imaging Ability. DNA Cell Biol. 2020;39(5):900-908. doi:10.1089/dna.2019.5057(IF:3.314)
[28] Ni XR, Zhao YY, Cai HP, et al. Transferrin receptor 1 targeted optical imaging for identifying glioma margin in mouse models. J Neurooncol. 2020;148(2):245-258. doi:10.1007/s11060-020-03527-3(IF:3.267)
[29] Feng H, Tang J, Zhang P, Miao Y, Wu T, Cheng Z. Anti-adipogenic 18,19-seco-ursane stereoisomers and oleane-type saponins from Ilex cornuta leaves. Phytochemistry. 2020;175:112363. doi:10.1016/j.phytochem.2020.112363(IF:3.044)
[30] Zhao Y, Li T, Tian S, et al. Effective Inhibition of MYC-Amplified Group 3 Medulloblastoma Through Targeting EIF4A1. Cancer Manag Res. 2020;12:12473-12485. Published 2020 Dec 3. doi:10.2147/CMAR.S278844(IF:2.886)
Payment & Security
Your payment information is processed securely. We do not store credit card details nor have access to your credit card information.
Inquiry
You may also like
FAQ
The product is for research purposes only and is not intended for therapeutic or diagnostic use in humans or animals. Products and content are protected by patents, trademarks, and copyrights owned by Yeasen Biotechnology. Trademark symbols indicate the country of origin, not necessarily registration in all regions.
Certain applications may require additional third-party intellectual property rights.
Yeasen is dedicated to ethical science, believing our research should address critical questions while ensuring safety and ethical standards.